Politics
Suvretta Capital Increases Stake in ArriVent BioPharma by 8.5%

Suvretta Capital Management LLC has expanded its investment in ArriVent BioPharma, Inc. (NASDAQ: AVBP) by 8.5% during the third quarter of 2023. According to its latest disclosure with the U.S. Securities and Exchange Commission (SEC), the institutional investor acquired an additional 290,664 shares, bringing its total holdings to 3,703,452 shares. This investment now constitutes approximately 1.7% of Suvretta’s overall portfolio, ranking as its 17th largest position.
As of its most recent filing, Suvretta Capital’s stake in ArriVent is valued at about $68.3 million, representing 8.97% of the company’s equity. Other institutional investors have also shown interest in ArriVent, contributing to a dynamic investment landscape for the biopharmaceutical firm.
Bessemer Group Inc. recently initiated a stake in ArriVent BioPharma worth approximately $26,000. Meanwhile, Russell Investments Group Ltd. reported a staggering increase in its holdings, boosting its investment by 9,747.6% in the same quarter. After acquiring an additional 2,047 shares, Russell now owns 2,068 shares valued at around $38,000.
Additionally, BNP Paribas Financial Markets has raised its position by 31.0%, now holding 2,571 shares valued at $56,000. Tower Research Capital LLC significantly increased its stake by 571.6%, owning 6,669 shares worth $145,000 after purchasing an additional 5,676 shares. Lastly, Jane Street Group LLC entered the market with a new stake valued at about $189,000. Collectively, hedge funds and institutional investors control approximately 9.48% of ArriVent’s stock.
Analyst Ratings and Market Performance
Several research firms have recently evaluated ArriVent BioPharma. BTIG Research initiated coverage on the company, issuing a “buy” rating with a target price of $42.00. Truist Financial also began coverage, setting a price objective of $43.00. Conversely, Weiss Ratings maintained a “sell (d-)” rating, indicating cautious sentiment towards the stock.
Analysts have mixed views, with two assigning a “Strong Buy” rating and eight issuing a “Buy” rating. One analyst has rated it as “Hold” and another as “Sell.” According to MarketBeat, ArriVent currently holds an average rating of “Moderate Buy” with an average target price of $40.78.
On the trading front, shares of ArriVent BioPharma experienced a slight decline of 0.8%, opening at $23.90 on the last trading day. The company boasts a market capitalization of $1.06 billion and a price-to-earnings (P/E) ratio of -5.42. Over the past year, the stock has fluctuated between a low of $15.47 and a high of $27.22, with a 50-day moving average of $22.60 and a 200-day moving average of $21.09.
ArriVent BioPharma last reported its quarterly earnings on March 5, 2024, revealing a loss of ($0.78) per share, which exceeded the consensus estimate of ($0.87) by $0.09. Analysts predict that ArriVent will post an average earnings per share of ($2.74) for the current fiscal year.
Company Overview
ArriVent BioPharma operates as a clinical-stage biopharmaceutical company focusing on the development of treatments for unmet medical needs, particularly in oncology. The firm specializes in targeted therapies for non-small cell lung cancer (NSCLC) and other solid tumors. Notably, it is developing Furmonertinib, a third-generation tyrosine kinase inhibitor currently undergoing multiple clinical trials for various epidermal growth factor receptor mutations (EGFRm) in NSCLC. This includes a phase 3 clinical trial for metastatic EGFRm NSCLC and ongoing phase 1b trials for other mutations.
ArriVent BioPharma’s ongoing research and investment interest underscore the growing focus on innovative cancer treatments, positioning the company as a key player in the biopharmaceutical sector.
-
 Science5 months ago
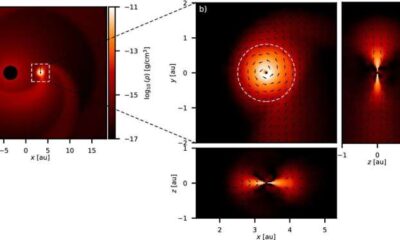
Science5 months agoALMA Discovers Companion Orbiting Giant Star π 1 Gruis
-

 World5 months ago
World5 months agoF-22 Raptor vs. Su-57 Felon: A 2025 Fighter Jet Comparison
-

 Science5 months ago
Science5 months agoUniversity of Hawaiʻi Joins $25.6M AI Project for Disaster Monitoring
-

 Politics3 months ago
Politics3 months agoU.S. Visa Rescheduling Hits H‐1B Applicants as New Vetting Rules Take Effect
-
 Science5 months ago
Science5 months agoOhio State Study Uncovers Brain Connectivity and Function Links
-

 Science6 months ago
Science6 months agoInnovator Captures Light at 2 Billion Frames Per Second
-

 Politics5 months ago
Politics5 months agoHamas Chief Stresses Disarmament Tied to Occupation’s End
-

 Entertainment5 months ago
Entertainment5 months agoMegan Thee Stallion Exposes Alleged Online Attack by Bots
-

 Entertainment5 months ago
Entertainment5 months agoPaloma Elsesser Shines at LA Event with Iconic Slicked-Back Bun
-

 Science6 months ago
Science6 months agoResearchers Challenge 200-Year-Old Physics Principle with Atomic Engines
-

 World5 months ago
World5 months agoPrince Andrew Faces Fallout from Scandals and Allegations
-

 Top Stories5 months ago
Top Stories5 months agoUrgent: Flight Cancellations Loom at Texas Airports Amid Shutdown









