Science
Yale Researchers Unveil New Synthesis Method for Anticancer Compound
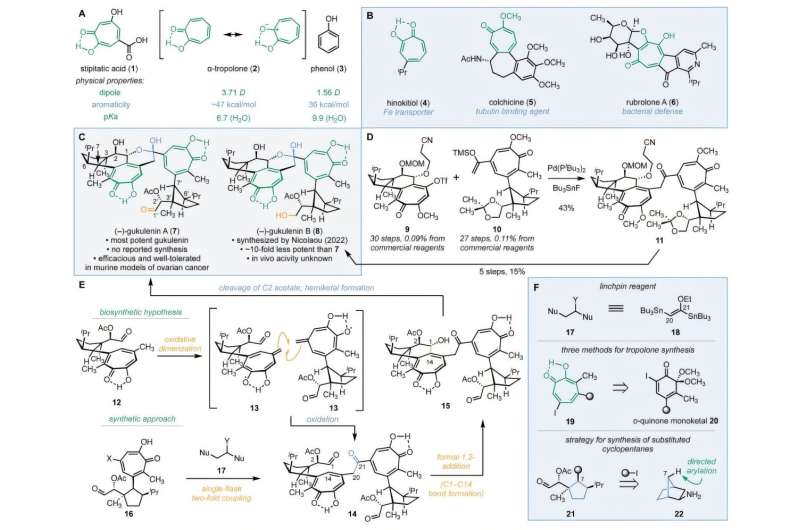
A team of researchers from Yale University has achieved a breakthrough in the synthesis of the complex natural product known as (–)-gukulenin A. This compound demonstrates significant cytotoxicity against ovarian cancer, with studies indicating it can reduce tumor size by more than 92% in mouse models. The findings were published on November 18, 2025, in the journal Science.
The synthesis of (–)-gukulenin A has long posed challenges for chemists due to its intricate molecular structure. It contains two α-tropolones, which are seven-membered aromatic rings characterized by strong molecular dipoles, alongside 10 precise stereocenters and sensitive chemical groups. Researchers tackled these complexities by employing a novel three-component assembly strategy inspired by biosynthetic pathways.
Innovative Synthesis Strategy
The Yale team implemented a three-step approach to synthesize (–)-gukulenin A. Initially, they constructed the two halves of the molecule, followed by a joining process, and finally closed the delicate rings. For the first half, they used readily available exo-2-norbornylamine, a rigid bicyclic molecule that guided the three-dimensional arrangement of the compound.
The researchers then developed a new ring-expansion method to convert a six-membered ring into the required seven-membered tropolone structure. The two monomers were joined using a newly created two-carbon linking reagent, (E)-1,2-di(tributylstannyl)-1-ethoxyethylene. The final step involved heating the intermediate to 120 °C, which triggered a precise reaction to close the fragile hemiketal ring.
After successfully synthesizing (–)-gukulenin A, the team designed 15 additional derivatives to investigate their structural features and cytotoxicity. Testing across four human cancer cell lines—lung, colon, leukemia, and ovarian—revealed that the derivatives with dimeric α-tropolones exhibited at least 10-fold greater potency than their monomeric counterparts. In some cases, they were found to be up to 200 times more effective.
Potential for New Therapeutics
The ability to synthesize (–)-gukulenin A at scale may pave the way for new therapeutic options for ovarian cancer. Notably, the compound has shown remarkable selectivity and tolerance in animal studies, setting it apart from many broadly cytotoxic natural products.
Researchers believe the cytotoxic potency of (–)-gukulenin A may stem from the α-tropolones’ affinity for divalent metals, enabling the compound to bind to two separate metal-containing proteins simultaneously. This discovery could help identify specific targets for (–)-gukulenin A and establish a foundation for preclinical evaluations of its synthetic derivatives as potential anticancer agents.
The research was led by Vaani Gupta and reflects a significant advancement in synthetic chemistry and cancer treatment options. As the scientific community continues to explore the therapeutic potential of compounds like (–)-gukulenin A, the implications for cancer therapies could be substantial.
This article has been fact-checked and reviewed to ensure accuracy and credibility, reflecting the rigorous editorial standards of the scientific community.
-
 Science1 month ago
Science1 month agoOhio State Study Uncovers Brain Connectivity and Function Links
-

 Politics1 month ago
Politics1 month agoHamas Chief Stresses Disarmament Tied to Occupation’s End
-

 Entertainment1 month ago
Entertainment1 month agoMegan Thee Stallion Exposes Alleged Online Attack by Bots
-

 Science4 weeks ago
Science4 weeks agoUniversity of Hawaiʻi Joins $25.6M AI Project for Disaster Monitoring
-

 Science2 months ago
Science2 months agoResearchers Challenge 200-Year-Old Physics Principle with Atomic Engines
-

 Entertainment1 month ago
Entertainment1 month agoPaloma Elsesser Shines at LA Event with Iconic Slicked-Back Bun
-

 World1 month ago
World1 month agoFDA Unveils Plan to Cut Drug Prices and Boost Biosimilars
-

 Top Stories1 month ago
Top Stories1 month agoFederal Agents Detain Driver in Addison; Protests Erupt Immediately
-

 Business1 month ago
Business1 month agoMotley Fool Wealth Management Reduces Medtronic Holdings by 14.7%
-

 Entertainment1 month ago
Entertainment1 month agoBeloved Artist and Community Leader Gloria Rosencrants Passes Away
-

 Politics1 month ago
Politics1 month agoNHP Foundation Secures Land for 158 Affordable Apartments in Denver
-
 Science2 weeks ago
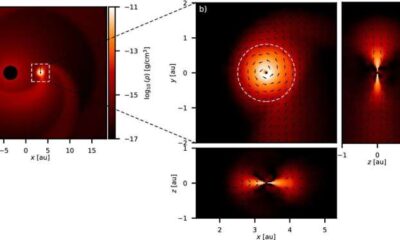
Science2 weeks agoALMA Discovers Companion Orbiting Giant Star π 1 Gruis









