Politics
FDA Proposes Increased Oversight of Vaccine Trials Following Memo

The Food and Drug Administration (FDA) is seeking increased oversight of vaccine trials and approvals following an internal memo that links the deaths of at least 10 children to the COVID vaccine. This memo, obtained by The New York Times, has raised significant concerns regarding vaccine safety and regulatory standards.
The memo, written by Dr. Vinay Prasad, director of the FDA’s vaccine division, outlines findings from a review of reports concerning childhood deaths attributed to myocarditis, an inflammation of the heart muscle. It suggests that these fatalities may be associated with the COVID vaccine, but lacks detailed information such as the ages of the children involved or their underlying health conditions. Importantly, the memo has not been published in a peer-reviewed medical journal, leading to skepticism among critics of Robert F. Kennedy Jr., the current Health Secretary, who has publicly questioned the safety of COVID vaccines despite the prevailing scientific consensus affirming their safety.
In his memo, Prasad stated, “This is a profound revelation. For the first time, the U.S. FDA will acknowledge that COVID-19 vaccines have killed American children.” He has proposed a range of new oversight measures and reviews of vaccine protocols, although it remains unclear if the White House has been informed of the memo’s contents. Potential changes could face scrutiny from lawmakers and pharmaceutical companies, complicating the regulatory landscape.
The memo follows a pattern of skepticism towards vaccination from federal authorities under Kennedy’s leadership. His administration has implemented policies restricting access to COVID vaccines for individuals under the age of 65 unless they have existing health conditions. Additionally, he has called for further studies on vaccines previously deemed safe, raising alarms among public health experts.
Health officials from both the Trump and Biden administrations have historically endorsed COVID vaccines as essential public health measures. According to the American Academy of Pediatrics, approximately 2,100 children have died from COVID-19 since the onset of the pandemic. This statistic underscores the urgency of maintaining public trust in vaccination efforts, particularly as cases of measles and whooping cough rise in the United States.
The timing of Prasad’s memo is notable, occurring just before a crucial meeting of the Centers for Disease Control and Prevention (CDC) vaccine committee. This committee, which includes members aligned with the so-called medical freedom community, is set to discuss the immunization schedule for children and the hepatitis B vaccine for newborns. Critics, such as Michael Osterholm, an infectious disease expert at the University of Minnesota, argue that the memo’s release was strategically timed to influence the committee’s discussions.
Prasad proposed several changes to vaccine oversight. These include ensuring that studies involving vaccines or placebos assess all demographic subgroups, such as pregnant women. He also criticized the current flu vaccine updating process as a “catastrophe of low-quality evidence,” indicating that this procedure would be reevaluated. Furthermore, he called for larger studies to establish the safety of administering multiple vaccines simultaneously, such as the flu and COVID vaccines.
These proposed changes could significantly increase costs for vaccine manufacturers, who are required to submit their studies to the FDA for evaluation. Public health experts warn that high-profile claims suggesting vaccines are unsafe could undermine public confidence, particularly as vaccination rates drop and preventable diseases resurface.
Dr. Paul Offit, a vaccine expert at the Children’s Hospital of Philadelphia, criticized the memo for lacking crucial context, such as the comparative deaths from COVID-19 among vaccinated and unvaccinated children. He described the document as an example of “science by press release,” highlighting the potential dangers of miscommunication in public health messaging.
Prasad’s memo follows a recent communication urging FDA staff to avoid publishing “obviously erroneous work,” emphasizing the importance of research aligned with the agency’s objectives. His leadership style has drawn mixed reactions, and he expressed frustration over perceived leaks of sensitive information regarding the agency’s direction and policies.
As the FDA navigates these complex issues, the implications of this memo could have far-reaching effects on vaccination policy and public health strategies in the United States.
-
 Science1 month ago
Science1 month agoOhio State Study Uncovers Brain Connectivity and Function Links
-

 Politics1 month ago
Politics1 month agoHamas Chief Stresses Disarmament Tied to Occupation’s End
-

 Entertainment1 month ago
Entertainment1 month agoMegan Thee Stallion Exposes Alleged Online Attack by Bots
-

 Science4 weeks ago
Science4 weeks agoUniversity of Hawaiʻi Joins $25.6M AI Project for Disaster Monitoring
-

 Science2 months ago
Science2 months agoResearchers Challenge 200-Year-Old Physics Principle with Atomic Engines
-

 Entertainment1 month ago
Entertainment1 month agoPaloma Elsesser Shines at LA Event with Iconic Slicked-Back Bun
-

 World1 month ago
World1 month agoFDA Unveils Plan to Cut Drug Prices and Boost Biosimilars
-

 Top Stories1 month ago
Top Stories1 month agoFederal Agents Detain Driver in Addison; Protests Erupt Immediately
-

 Business1 month ago
Business1 month agoMotley Fool Wealth Management Reduces Medtronic Holdings by 14.7%
-

 Entertainment1 month ago
Entertainment1 month agoBeloved Artist and Community Leader Gloria Rosencrants Passes Away
-

 Politics1 month ago
Politics1 month agoNHP Foundation Secures Land for 158 Affordable Apartments in Denver
-
 Science2 weeks ago
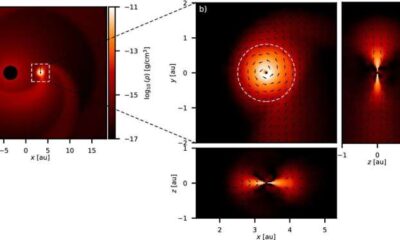
Science2 weeks agoALMA Discovers Companion Orbiting Giant Star π 1 Gruis









